By: Eric Watson (2017-18 Hagenah Postdoctoral Fellow)
The ability of a single genome to produce two different sexes is as fascinating as it is mysterious. In species with sex chromosomes, the difference in the genetic makeup between the sexes is typically limited to a few genes on the Y (or W) chromosome. Because the vast majority of genes are shared between males and females, sex differences are largely accomplished by altering the way in which the genes are regulated between the sexes. It’s not what you have, but how you use it.
In the relatively few species investigated (e.g. humans, mice, fruit flies), large portions of the genome are regulated differently between the sexes. These regions tend to cluster on the sex-chromosomes, resulting in patterns of overall feminization or masculinization of X (or Z) chromosomes depending on the species. Not only does this “sex-biased expression” contribute to sexual dimorphism, but it also contributes to differences in environmental stress tolerance and leads to sex-biases in many common diseases.

Me and Jane Pascar dissecting copepods. Jane learned micro dissections during her undergrad and was the inspiration for this project. Now she is moving on to a PhD at Syracuse University.
My work seeks to understand how sex-biased genes lead to differences in the ability of either sex to adapt to the environment, and live a long and healthy life by removing the biases that typically stand in the way of a generalized theory. To start, I ask: Why are sex chromosomes so important? Are there exceptions to the typical XY sex-determination system? As it turns out, many sex-determination systems have evolved in animals and plants, yet research is mostly biased towards XY systems.
Enter the splash-pool copepod, Tigriopus californicus affectionately called “Tigs”. These little (and I do mean little) critters are abundant in the rocky intertidal along the Pacific coast, and are an ideal organism to study topics like adaptation to environmental stress, longevity and aging, and the accumulation of genetic differences in isolated populations. They’re easy to find, collect, breed, and seem to be at home in a laboratory culture making them an ideal genetic model system. But, one trait makes them a game changer: they do not have sex chromosomes.
Instead of possessing an XY sex-determination system like flies, humans and mice, sex determination in T. californicus involves multiple chromosomes that operate in combination to produce males or females. We haven’t yet discovered which genes are involved.
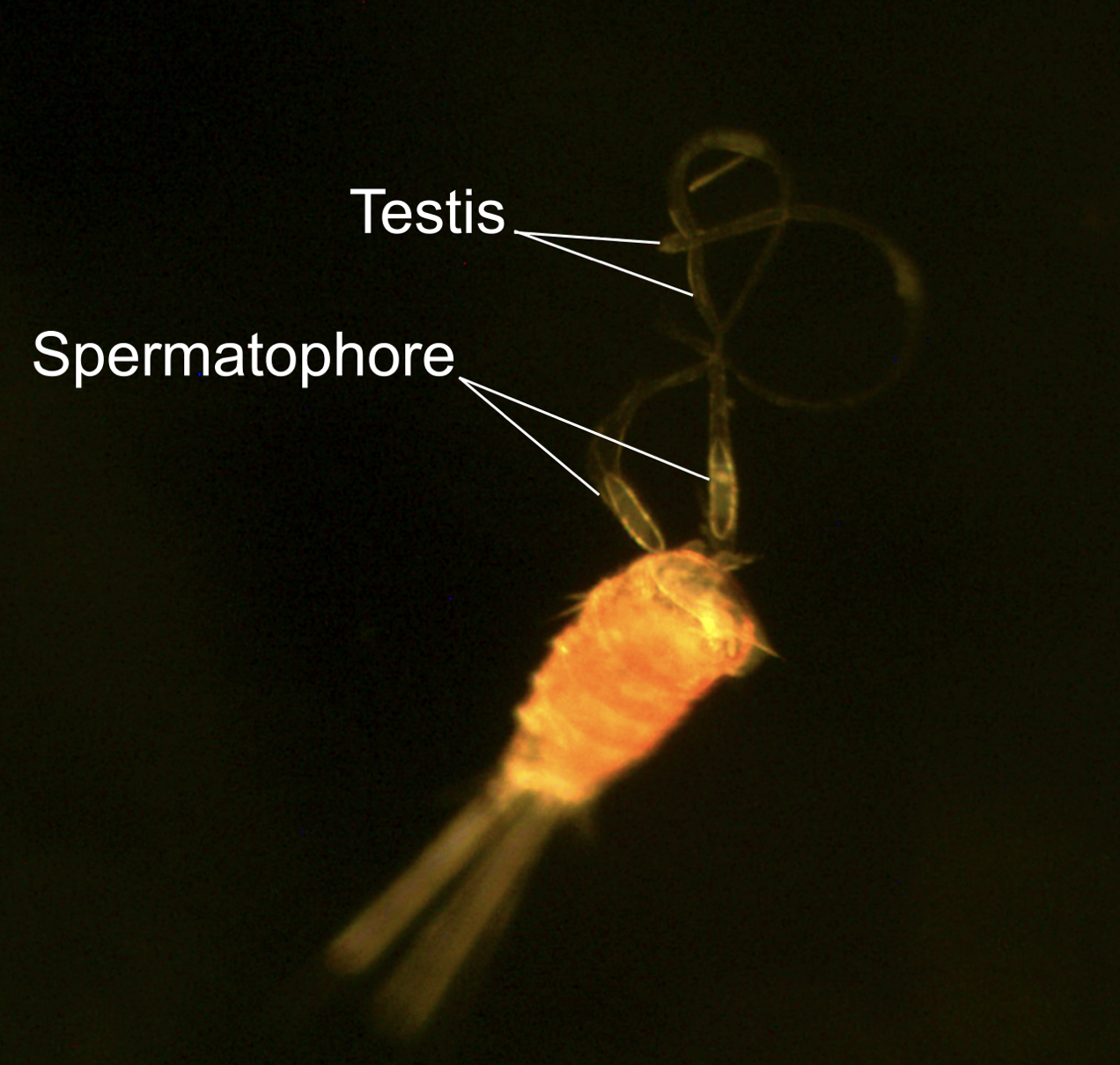
Eureka!! This partial dissection of a male copepod shows paired testes. Many species of copepods only have one testis and this is the first image describing paired testes in Tigriopus. Photo credit: Jane Pascar
How do sex differences evolve in species without sex chromosomes? To answer this question, I am beginning with a study to determine which genes are sex-biased and which genes are expressed solely in the male and female reproductive tracts. Identifying these genes will aid in discovering the genes responsible for sex-determination in T. californicus, and also test the prediction that they should be distributed randomly in the genome as opposed to clustered together as they are on the sex-chromosomes of other organisms. The evolutionary implications of heredity in the absence of sex chromosomes are wide-ranging yet understudied, but T. californicus brings promise as a genetic model system for evolutionary biology.

